Bohr's atomic model
The Rutherford's atomic model had some failures that cast doubt on it, for example, classic physics says that a charged particle release energy when it is moving, so, an electron will release energy meanwhile is moving around nucleus, that is to say, the electron will fall into nucleus, destroying the atom, due to the electrostatic force between nucleus and electron.
To solve this problem, Niels Bohr, applied the Max Plack's quantum theory (which said that energy was "quantized", that is to say, that energy was absorbed or released by small "bags", called quanta), to the Rutherford's atomic model, making a new one called the stationary atomic model. Its main principles were the following:
- The electron's energy is quantized
- The electron moves around the nucleus into a stationary energy level (SEL), where it will not lose or gain energy. This level will be as close to the nucleus as possible.
- When an electron gains energy, it moves to a higher SEL. When the electron returns to its basal SEL, it will release the excees of energy as light.
- The electron makes circular orbits around the nucleus.
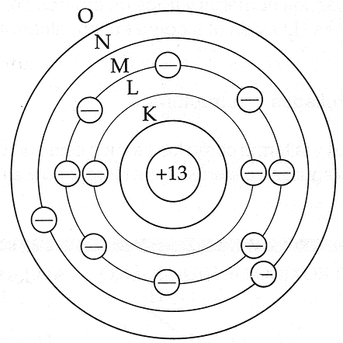
Bohr stablished that the orbits will be represented by letters (from K to Z), where K has the less energy, as its shown in Image 6.
Unfortunately, this model only worked on Hydrogen-type atoms, but with polielectronic atoms, it only could determine the maximum quantity of electrons an energetic level could have.
Image 6: Bohr's model (got from http://www.pps.k12.or.us)