Introduction
A little bit of history
First Approaching
Although chemistry is considered a science from 19th century approximately, the first sorties that humankind made into this area were made even before any grat civilization could be established on Earth. We would be able to start saying that the fact of the human's appearence on our planet could be taken as the first approaching of man to chemistry. But, most of chemists think that the real first approaching occured when fire was discovered by Homo Erectus (accidentally).
Posterior, human being begins to venture into metallurgical industry, starting with Copper, later mixing this metal with Tin to make an alloy named Bronze, to finish creating furnitures made with Iron.
From Democritus to Dalton
On 5th century BC, Ancient Greek philosophers were divided into 2 main ideas about the Universe conformation: the atomist theory and the four elements theory.
The former, was postulated by Leucippus and Democritus. This theory said that the Universe was formed by very small particles called atoms (from greek a = without, tomo = division). These particles were eternal, indivisibles and indestructible, which mixture yielded the matter that exists in the Universe. These particles moved through vaccum and this one was cappable to separate the matter. The order, shape and size of atoms produced differencies between one substance and another.
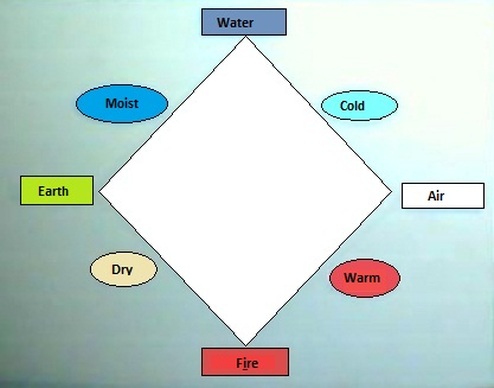
The second theory was a mixture of ideas from many philosophers, just like Aristotle and Thales of Miletus. This theory said that matter could be divided inifinite times, that is to say, they didn't believe in a inicial particle, just as said the theory explained before. These philosophers thought that matter contained on Earth was made from 4 elements: Fire, Water, Wind and Earth; and that the diferents combination of them gave the matter diferents characteristics, for example, a featherhad high percentages of air and earth, but mostly had earth, so a feather will fall to the earth but softly, because it had a high percentage of air. The Universe was made by a fifth element, called Ether, which was a perfect substance. The four elements and their combination are shown in Image 1.
Image 1: The four elements and their intermidate states.
This theory had a great acceptance, and this meant that it ruled Europe for almost 2,000 years, but before seeing what happened then, let's see what happened on those 2 millennia.
The Alchemists
After Western Roman Empire had felt, the territories began to take more protagonism, same thing that happened with Catholic Church. At this time, a new thought appeared in China called alchemy. The alchemists are mainly known for their interest in turning every metal into gold, which ona was considered as a divine metal for the Church. It's also known that they ate gold to obtain divine inmortality. But, despite of all these things that can sound fantastic, the alchemists are considered as "the chemistry precursors", because they reached very important knowledgements in some parts of chemistry, just as Paracelso, who is considered the father of the iatroquímica (medical chemistry), which is the origin of the actual pharmacochemistry.
The Flogist and Lavoisier
On 19th century, J.J. Beecher postulated the flogist theory. According to this theory, the matter had a intrinsic substance called flogist, which go out the matter when it oxidizes, and go in the matter when it reduces (reduction-oxidation reactions will be seen later). Beecher said that, if his theory was true, and considering the fact that a oxidized substance has ore weight than the original substance, the flogist must have a negative weight, that is to say, the more flogists left a substance, more weight it had. This theory was accepted for 75 years, aproximately, until Antoine Lavoisier demostrated how wrong Beecher was, making an experiment in which an oxidized substance was put into a flask without air. If Beecher was right, the substance had to stay oxidized, because there was not any other substance that could give flogist. But that didn't occur, because the oxidized substace change into its reduced form, leaving a gas, that Lavoisier called oxigen. This and many other contributions gave him the title of father of chemistry.
The atom is back
While first years of 19th century were passing by, John Dalton, an english chemist and physicist, took the ideas of Democritus and formulated what is considered the first atomic model of the history. Some of his postulates were:
- Substances are formed by small, indestrustible and indivisible particles named atoms.
- The atoms of a substance have all same shape and size (are equals), but they differ from those who came from another substance.
- The atoms mix with other atoms to form compounds, in small and simple proportions.
- The atoms can combine in more than one small and simple proportion.
This theory ruled the world for almost one century, but during this time some doubts that this theory cannot explain were appearing. But before seeing that, come to learn some importants basic concepts about chemistry that will help you to get a better understanding of this science.