Thomson's Atomic Model
Thomson and the electron:
When the 19th century came to an end, scientists began to see unsusual behaviours in matter, which cannot be explained bye the atomic model proposed by Dalton, for example, the Millikan's experiment at the middle of 19th century, which consisted in dropping an oil drop through an electric field into a vacuum-packed tube. This experiment could determine that the minimun charge which was acquired by the oil drop was equal to 1,6x10-19 C. This and many others electromagnetic experiencies gave an idea that the "indivisible spherical" model thought by Dalton was wrong and a charged particle, which could be affected by changes in the electric field, was present.
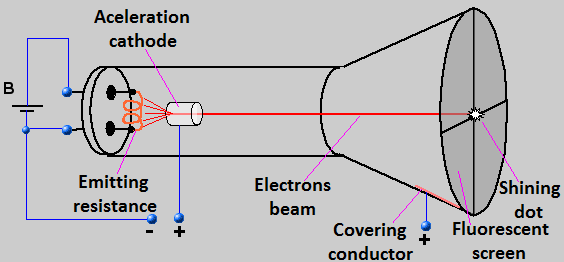
After those discoveries, scientists tried to give some explanations about those phenomena. The most important explanation was made by Thomson at the end of 19th century and beginning of the 20th, which consisted in the use of the "cathode ray tube" (Image 2), that was formed by two electrodes connected to a high voltage source (HVS) and to a vacuum-packed tube. When an electric current passed from the HVS to the anode (negative electrode), a luminous ray was seen going from the anode to the cathode (postive electrode). This ray was called cathode ray and was studied by Thomson, who applied a electromagnetic field to this ray, and saw that the ray was deviated from its rect line to the positive side of the field, that plus the knowledge that two opposite charges will atract each other, gave him the conclusion that the cathode ray was composed by negative particles coming from the anode, which he named electrons. After this discovering, Thomson formulated a new atomic model, which was called "raisin cake" (it is represented in an approximately way by Image 3), which said that electrons were set into a great positive-charged sphere with the same magnitude of charge (this was the first time someone spoke about atom's electroneutrality), and Thomson could determine that the quotient between the electron mass (discovered by Millikan) and its charge was equal to 1,756x108.
Image 2: Cathode ray tube (it was obtained from www.astronomos.net23.net and translated by webmaster)
Image 3: White balls represents the electrons and the red ball represent the positive-charged particle.